
Priscilla Perez da Silva Pereira1; Andriely Alayne Carvalho Sabini2; Rosa Maria Ferreira de Almeida2; Daniela Oliveira Pontes2; Márcia Maria Bezerra Mororó Alves3; Viviane Alves de Sousa3; Magzan da Silva Azevedo3; Adalgiza de Souza Botelho3; Surlange Freire Ramalhaes3; Edilson Batista da Silva3; Maria Artele da Gama Baldez3; Elizabeth Carmen Duarte1
DOI: 10.5935/0004-2749.2025-0175
ABSTRACT
PURPOSE: Endophthalmitis is one of the most important adverse events after cataract surgery, as it can lead to total vision loss. This study aimed to describe the occurrence of endophthalmitis after phacoemulsification with intraocular lens implantation in patients treated in a community setting in Porto Velho, Rondônia, Brazil.
METHODS: This retrospective cohort study was conducted using a database of 649 medical records of patients who underwent surgery and were followed for three months. Poisson regression analysis was used to estimate relative risks and 95% confidence intervals (95% CIs).
RESULTS: The incidence of confirmed endophthalmitis was 11.94% (95% CI, 9.50-14.76), while the incidence of confirmed and probable cases was 20.50% (95% CI, 17.52-23.73). For confirmed cases, bilateral surgery and the use of lens model 3 were identified as risk factors for endophthalmitis, whereas age over 70 yr and preoperative antibiotic use were protective factors. For confirmed and probable cases, brown and yellow skin color, bilateral surgery, and the use of lens model 3 were also identified as risk factors. Gram-negative bacteria were the predominant etiological agents, and corneal edema was the main clinical manifestation. The mean duration of treatment was eight days, and 27.12% of patients used antibiotics.
CONCLUSION: The incidence observed was substantially higher than that reported in the literature, with a predominance of Gram-negative agents and an association with bilateral surgeries and the Eyeol intraocular lens model. These findings reinforce the need for continuous epidemiological surveillance and the implementation of specific biosafety and infection control protocols during cataract surgery campaigns.
Keywords: Endophthalmitis; Disease outbreaks; Phacoemulsification; Lens implantation, intraocular; Lenses, intraocular; Cataract; Risk factors; Anti-bacterial agents
INTRODUCTION
The prevalence of cataracts among individuals aged over 60 yr is estimated at 54.38% (95% confidence interval [95% CI], 47.57-61.18), and cataracts are responsible for up to 50% of cases of blindness worldwide(1). Cataracts are defined as any opacification of the lens, with or without a reduction in visual acuity. Factors such as advanced age, exposure to sunlight, comorbidities, previous eye diseases, ocular trauma, socioeconomic status, and geographic region may directly or indirectly influence cataract development(2).
To restore visual function in patients with senile cataracts, surgery is the only definitive treatment, as correction with eyeglasses provides only temporary benefit. Phacoemulsification combined with intraocular lens implantation is a safe and effective procedure that enables visual rehabilitation in most cases(1). One of the most serious complications of this procedure is endophthalmitis; however, it is considered rare. A study conducted in South Korea between 2014 and 2017 involving nearly one million patients reported an incidence of 0.063%(3). In the United States, an incidence of 0.04% was observed among more than eight million cataract surgeries(4). A retrospective cohort study conducted in Argentina between 2006 and 2020 reported an incidence of 0.10%(5). In Brazil, a study performed between 2008 and 2014 involving more than 27,000 cataract surgeries reported an incidence of 0.13%, with annual variation ranging from 0.04% to 0.27%(2). Another Brazilian study conducted in a northern state found that among 3,999 procedures performed during joint efforts in 2013 and 2014, 1.67% presented perioperative or postoperative complications(6).
In most cases, endophthalmitis is caused by bacteria originating from the palpebral margin and tear film of the patient's conjunctival flora(7). Other possible sources of infection include contamination of equipment, instruments, surgical supplies, solutions, surgical drapes, or intraocular lenses, as well as failures in adherence to good clinical practices(7). Clinical manifestations usually appear between one and seven days after surgery(8). The most common signs and symptoms include conjunctival hyperemia, hypopyon, vitreous opacity, anterior chamber reaction, corneal edema, and reduced visual acuity(8). Early recognition of endophthalmitis is essential, as severe cases may progress to blindness(9). In addition to visual impairment, endophthalmitis may negatively affect patients' functional capacity, mental health, and quality of life, and it increases healthcare costs for both patients and health systems(10).
To address the growing number of cataract cases, it is estimated that approximately 600,000 cataract surgeries are required annually in Brazil(11). To improve access, particularly in remote regions and in the context of budgetary and managerial limitations, joint surgical efforts (mutirões) are commonly implemented(6).Cataract prevention campaigns in Brazil began in the 1980s, with the National Cataract Campaign (2001-2006) representing a major milestone in expanding surgical access. Currently, federal funding, private contracts, and parliamentary amendments support cataract surgery within the Unified Health System(12-14).
Between February 14 and 23, 2022, a cataract surgery campaign was conducted in Porto Velho, the capital of Rondônia, in northern Brazil. On February 23, the first suspected case of endophthalmitis was identified during outpatient follow-up. On the same day, surgical activities were suspended by the company responsible for the campaign. Following an investigation by the State Center for Strategic Health Surveillance Information of Rondônia, an outbreak of endophthalmitis was confirmed. Therefore, this study aimed to describe the occurrence of endophthalmitis after phacoemulsification with intraocular lens implantation among patients treated during this joint effort in Porto Velho, Rondônia, Brazil.
METHODS
This retrospective cohort study was conducted using a database of 649 medical records of patients who underwent phacoemulsification with intraocular lens implantation and were followed for three months. Some patients underwent bilateral surgery, resulting in a total of 1,044 procedures performed over a 10-day period, with a mean of 104 procedures per day (minimum 90 and maximum 120).
Data were collected from medical records and included sociodemographic characteristics, preoperative examinations, medical history, materials and techniques used during surgery, postoperative signs and symptoms, and examinations and treatments performed after surgery. Not all collected variables were included in the present analysis; therefore, only those with greater statistical or epidemiological relevance were analyzed and presented.
A probable case was defined as a patient who underwent the procedure and presented at least two of the following signs or symptoms of infection: decreased visual acuity, ocular pain, corneal edema, conjunctival hyperemia, hypopyon, anterior chamber reaction, vitreous opacity, and/or receipt of postoperative intravitreal antimicrobial therapy(10). Patients with a positive microbiological culture from vitreous and/or aqueous humor samples were classified as confirmed cases(8).
Data were summarized using absolute and relative frequencies for categorical variables and measures of central tendency for numerical variables. Pearson's chi-square test or Fisher's exact test was used in the bivariate analysis, as appropriate. After bivariate analysis, covariates were assessed for multicollinearity, defined as correlations greater than 0.80. Poisson regression analysis was performed to estimate relative risks (RRs) and 95% confidence intervals (95% CIs). In the final model, variables with p-values <0.05, those that altered the association measure by at least 20%, or those that improved model fit were retained using a forward stepwise selection strategy. Statistical analyses were performed using Stata version 16.0 (College Station, Texas).
This study is part of the matrix project entitled Good Practices in Patient Care, Infection Control, and Processing of Health Products in the State of Rondônia and was approved by the Research Ethics Committee of the Universidade Federal de Rondônia (CAAE: 20070719.5.0000.5300). All data were fully anonymized before being provided to the researchers by the Rondônia State Health Surveillance team in April 2022.
RESULTS
A total of 649 medical records were analyzed. Of these, 446 patients did not present signs or symptoms suggestive of endophthalmitis, 70 were diagnosed with culture-confirmed endophthalmitis, and an additional 63 patients were classified as probable cases, as they presented at least two signs or symptoms suggestive of endophthalmitis. The incidence of confirmed endophthalmitis was 11.94 per 100 surgeries (95% CI, 9.50-14.76), while the combined incidence of confirmed and probable cases was 20.50 per 100 surgeries (95% CI, 17.52-23.73).
Half of the patients were female, most self-identified as having black, brown, or yellow skin color, and 35% were older than 70 yr (Table 1). Some patients had a history of previous ophthalmologic surgery (21.11%), used antibiotics before surgery (19.34%), and more than half underwent bilateral surgery on different days. In addition, 51.67% of patients received lens model 3. Statistically significant differences (p<0.05) were observed for confirmed cases with respect to age group, history of previous ophthalmologic surgery, bilateral surgery, and two lens models (Table 1). For confirmed and probable cases, skin color, history of previous ophthalmologic surgery, bilateral surgery, and three lens models showed statistically significant associations.
For confirmed cases, bilateral surgery increased the risk of endophthalmitis by 1.80 (95% CI, 1.03-3.17), and the use of lens model 3 was also identified as a risk factor (RR: 11.87; 95% CI, 3.71-37.98; Table 2). Age over 70 yr (RR: 0.50; 95% CI, 0.25-0.97) and preoperative antibiotic use (RR: 0.45; 95% CI, 0.21-0.95) were identified as protective factors. For confirmed and probable cases, brown and yellow skin color (RR: 1.64; 95% CI, 1.05-2.55), bilateral surgery (RR: 1.90; 95% CI, 1.23-2.92), and the use of lens model 3 (RR: 3.97; 95% CI, 2.18-7.25) were also associated with an increased risk of endophthalmitis.
The mean time between the date of surgery and the initiation of treatment for endophthalmitis was eight days, ranging from 1 to 38 days. Among the evaluated medical records, 97.69% indicated that patients attended a postoperative visit within 24 h after surgery, and 73.50% of these patients already presented signs of infection. A second postoperative visit was recorded for 60.40% of patients (Table 3). Although 37.44% of patients still showed signs of infection in the first operated eye, half of them scheduled an evaluation in preparation for surgery on the second eye. Signs of infection were observed in 32.36% of patients at the third postoperative visit, decreasing to 19.88% at the fourth visit and to 13.56% by the fifth and final postoperative appointment.
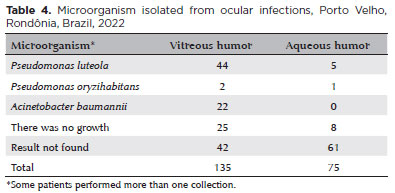
Biological material was collected in 21.91% of the evaluated cases. Three microorganisms were identified in vitreous and/or aqueous humor samples: Pseudomonas luteola, Pseudomonas oryzihabitans, and Acinetobacter baumannii (Table 4).
According to medical records, among the 649 patients, 12.17% used atropine, 14.79% prednisone, 17.57% vancomycin, 3.54% dexamethasone, and 14.95% amikacin, in addition to other medications that were recorded in fewer than 2% of cases.
DISCUSSION
In contrast to the global trend of decreasing incidence, the combined confirmed and probable incidence of postoperative endophthalmitis in Rondônia reached 20.50%, a rate substantially higher than those reported in the literature. This represents the largest outbreak of endophthalmitis after cataract surgery documented worldwide(3-5).
Three variables were identified as risk factors for endophthalmitis: bilateral surgery, skin color, and the use of lens model 3. Delayed sequential bilateral cataract surgery is often preferred by both surgeons and patients due to concerns regarding bilateral sequelae, including endophthalmitis and postoperative macular edema. In the present study, bilateral surgeries were performed on different days; nevertheless, an association between bilateral surgery and endophthalmitis was observed. This finding is consistent with a systematic review of 13 studies involving 11,068,622 individuals, which concluded that surgeries performed on different days do not protect against infection(15).
Regarding skin color, black, brown, and yellow skin were associated with an increased risk of confirmed and probable endophthalmitis compared with white skin. A study conducted in the United States between 2010 and 2014 involving more than 150,000 patients reported higher rates of endophthalmitis after cataract surgery among men, older individuals, African Americans, and Native Americans(3). The increased risk observed among Black and Native American populations may be related to social disadvantage and poorer overall health status in these groups(16).
Four intraocular lens models were used, all packaged in sterile vials and preserved in saline solution until use. All lenses were suitable for correcting myopia and hyperopia, differing mainly in their hydrophobic properties. Only the AcrySof IQ (Alcon) lens is hydrophobic, whereas the others are hydrophilic. Hydrophobic acrylic lenses have been shown to exhibit lower bacterial adhesion and are associated with a reduced risk of post-phacoemulsification endophthalmitis(17). A systematic review evaluating lens materials—including acrylic, hydrogel, polymethyl methacrylate (PMMA), and silicone—found no significant differences in endophthalmitis risk when PMMA was used as the reference material(18). No evidence was identified in the literature indicating an intrinsic increased risk associated with lens model 3. Therefore, contamination of the lens itself is unlikely, particularly as lenses from the same batch were used without reports of infection. A more plausible hypothesis is contamination during intraoperative handling.
A protective association was observed for age over 70 yr and preoperative antibiotic use among confirmed cases. Although higher rates of endophthalmitis are generally expected in older populations(18), the protective association observed in this study may be related to the presence of a home caregiver during the postoperative period.
Several studies have demonstrated that endophthalmitis rates decrease with the use of antibiotic prophylaxis, although variability remains due to differences in patient characteristics and risk factors(19). Preventive measures include the use of 0.66% povidone-iodine eye drops before surgery(20) and the administration of intracameral antibiotics during the procedure(21,22). In addition to patient-level interventions, infection prevention measures must also be implemented in operating rooms and surgical instrument processing, including air filtration review, individual use of eye drops per patient, frequent replacement of povidone-iodine solutions, increased availability of phacoemulsification handpieces, and regular review of sterilization protocols(5).
Postoperative endophthalmitis is most commonly caused by Gram-positive bacteria, particularly Staphylococcus species, followed by Gram-negative bacteria and, less frequently, fungi(23). A systematic review of studies conducted in India identified Staphylococcus species as the most common pathogens, followed by Gram-negative Pseudomonas species(24). In contrast, this study found a predominance of Gram-negative bacteria, particularly Pseudomonas luteola and Acinetobacter baumannii, in vitreous and/or aqueous humor samples. Similar findings have been reported in studies conducted in Asia(9,25) and Argentina, where Pseudomonas aeruginosa accounted for 45% of cases(5). Endophthalmitis caused by P. luteola is rare, with the first reported case described in the Philippines in 2007(26).
Although P. aeruginosa is typically associated with more severe cases, P. luteola has been linked to a more favorable prognosis. This organism is commonly found in water, soil, and humid environments and predominantly affects immunocompromised individuals or those using invasive devices(27). In hospital settings, P. luteola may be present in high-transmission areas, such as alcohol dispensers(28).
Only three previous studies have reported endophthalmitis caused by Acinetobacter baumannii(29). This pathogen is characterized by intrinsic antibiotic resistance and prolonged survival on surfaces, limiting therapeutic options and facilitating nosocomial transmission(30). Its ability to colonize skin, medical devices, and the respiratory tract of patients and healthcare workers has been implicated in epidemic outbreaks(31).
All patients were instructed to attend a postoperative visit within 24 h after surgery. Corneal edema was the most common clinical manifestation, observed in 68.26% of patients at the first postoperative visit, followed by hypopyon, decreased visual acuity, conjunctival hyperemia, ocular pain, and anterior chamber reaction. Ocular pain and hypopyon have been reported in up to 75% of endophthalmitis cases in other studies(9,25).
The mean interval between surgery and initiation of treatment was eight days, consistent with previous reports(32). During this period, prompt access to medical care is essential(4). As most complications occur in the late postoperative period, active follow-up of patients is critical, even during large-scale surgical campaigns, which is not always consistently implemented in Brazil(6).
Limitations of this study include the inability to verify potential sources of infection, such as surgical instruments, intraocular lenses, environmental surfaces, or other materials. In addition, the data were obtained from medical records completed by professionals hired for the surgical campaign, which may have resulted in incomplete information or imprecise case definitions, for example, due to the lack of requests for microbiological culture tests. Another limitation was the inability to assess how postoperative care was provided by patients in their home environments.
The occurrence of microbial contamination in Porto Velho may be associated with multiple factors. One possible cause is the large number of surgeries performed over a short period in adapted facilities, a limitation that has been highlighted as a challenge in cataract surgery campaigns(11). Furthermore, a systematic review reported that the reuse of surgical instruments in ophthalmology to reduce costs is frequently associated with infections. Another systematic review identified contamination of intraocular solutions, in addition to surgical instruments, as major sources of infectious transmission in cataract surgery(32). Other studies have also reported contamination of ocular viscoelastic devices(33), surgical vials(34), and prefilled saline syringes(35) as factors associated with endophthalmitis. Additional contributing factors may include surgeon experience, surgical volume, and workflow errors, particularly inadequate preparation and processing of surgical instruments(8,10). Based on the findings of this study, we believe that the outbreak of endophthalmitis in Porto Velho may have been related to inadequate hygiene practices involving surfaces, equipment, and materials used during the procedures.
As this represents the largest outbreak of endophthalmitis ever reported worldwide, these results highlight the urgent need for specific protocols for contracting, performing, and monitoring community-based cataract surgery services, which are commonly implemented, particularly in regions with limited access to healthcare. Such protocols should enable healthcare professionals to adopt good clinical practices, including appropriate cleaning and disinfection of surfaces, equipment, and surgical materials.
AUTHORS' CONTRIBUTIONS:
Significant contribution to conception and design: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes. Data Acquisition: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes, Márcia Maria Bezerra Mororó Alves, Viviane Alves de Sousa, Magzan da Silva Azevedo, Adalgiza de Souza Botelho, Surlange Freire Ramalhaes, Edilson Batista da Silva, Maria Artele da Gama Baldez. Data Analysis and interpretation: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes, Elisabeth Carmen Duarte. Manuscript Drafting: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes, Márcia Maria Bezerra Mororó Alves, Viviane Alves de Sousa, Magzan da Silva Azevedo, Adalgiza de Souza Botelho, Surlange Freire Ramalhaes, Edilson Batista da Silva, Maria Artele da Gama Baldez, Elisabeth Carmen Duarte. Significant intellectual content revision of the manuscript: Elisabeth Carmen Duarte. Final approval of the submitted manuscript: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes, Márcia Maria Bezerra Mororó Alves, Viviane Alves de Sousa, Magzan da Silva Azevedo, Adalgiza de Souza Botelho, Surlange Freire Ramalhaes, Edilson Batista da Silva, Maria Artele da Gama Baldez, Elisabeth Carmen Duarte. Statistical analysis: Priscilla Perez da Silva Pereira, Andriely Alayne Carvalho Sabini, Rosa Maria Ferreira de Almeida, Daniela Oliveira Pontes, Elisabeth Carmen Duarte. Obtaining funding: not applicable. Supurvision of Administrative, technical, or material support: Rosa Maria Ferreira de Almeida. Research group leadership: Priscilla Perez da Silva Pereira.
REFERENCES
1. Teles LP, Passos MA, Teles CO, Lima SO. Analysis of quality of life before and after cataract surgery with intraocular lens implantation. Rev Bras Oftalmol. 2020;79(4):242-7.
2. Luz RA, Dall'Oglio LPS, Silva FS, Ghirelli W, Padoveze MC. Endophthalmitis after cataract surgery: results from seven years of epidemiological surveillance. Rev Bras Oftalmol. 2019;78(2):86-90.
3. Kim SH, Yu MH, Lee JH, Kim SW, Rah SH. Endophthalmitis after cataract surgery in Korea: a nationwide study evaluating the incidence and risk factors in a Korean population. Yonsei Med J. 2019;60(5):467-73.
4. Pershing S, Lum F, Hsu S, Kelly S, Chiang MF, et al. Endophthalmitis after Cataract Surgery in the United States: A Report from the Intelligent Research in Sight Registry, 2013-2017. Ophthalmology. 2020;127(2):151-8.
5. Segretín Gutiérrez EFE, García MM, Bursztyn M, Benavente Defferrari MM, Ortiz-Basso T. Incidence of endophthalmitis post cataract surgery in a Tertiary Hospital of Buenos Aires. Medicina (B Aires). 2022;82(6):851-855.
6. Meirelles MG, Santana TS, Vieira LT, Costa CS, Celestino KA, Abud MB, Ávila MP. Prevalence of cataract surgery complications in a health care campaign. Braz J Develop. 2020;6(7):53783-90.
7. Simina DS, Larisa I, Otilia C, Ana Cristina G, Liliana MV, Aurelian MG. The ocular surface bacterial contamination and its management in the prophylaxis of post-cataract surgery endophthalmitis. Rom J Ophthalmol. 2021;65(1):2-9.
8. Agência Nacional de Vigilância Sanitária (ANVISA). Medidas de prevenção de endoftalmites e de síndrome tóxica do segmento anterior relacionadas a procedimentos oftalmológicos invasivos. Brasília (DF): ANVISA; 2017 [citado 2026 Jan 30]. Disponível em: file:///Users/ednatrother/Downloads/Medidas%20de%20Preven%C3%A7%C3%A3o%20de%20Endoftalmites%20-%20Caderno%209.pdf
9. Sun J, Guo Z, Li H, Yang B, Wu X. Acute infectious endophthalmitis after cataract surgery: epidemiological characteristics, risk factors, and incidence trends, 2008-2019. Infect Drug Resist. 2021;14:1231-8.
10. Schmier JK, Hulme-Lowe CK, Covert DW, Lau EC. An updated estimate of costs of endophthalmitis following cataract surgery among Medicare patients: 2010-2014. Clin Ophthalmol. 2016;10:2121-7.
11. Kara-Junior N. Are cataract surgery campaigns necessary for preventing blindness in developing countries? Arq Bras Oftalmol. 2025;88(4):e20241016.
12. Kara-Junior N, Rossi S. Epidemiology of Cataract-Related Blindness in Brazil: 30 Years of Public Policy Evolution: A Review Article. Am J Ophthalmol. 2025;273:205-11.
13. Kara-Junior N, Almeida HG, Jorge PA, Carricondo PC, Kara-José N. Consequences of cataract surgery public policies run by private contractors. Arq Bras Oftalmol. 2017;80(1):V-VI.
14. Rossi S, Jorge PA, Scherer R, Kara-Junior N. Progression in the Number of Cataract Surgeries in Brazil: 10 Years of Evolution. Ophthalmic Epidemiol. 2025;32(3):285-92.
15. Aiello F, Gallo Afflitto G, Leviste K, Swaminathan SS, Yoo SH, Findl O, et al. Immediate sequential vs delayed sequential bilateral cataract surgery: systematic review and meta-analysis. J Cataract Refract Surg. 2023;49(11):1168-79.
16. Keay L, Gower EW, Cassard SD, Tielsch JM, Schein OD. Postcataract surgery endophthalmitis in the United States: analysis of the complete 2003 to 2004 Medicare database of cataract surgeries. Ophthalmology. 2012;119(5):914-22.
17. Qiu X, Wu Y, Jiang Y, Ji Y, Zhu X, Yang J, et al. Management and Microbiological Characteristics of Membrane Formation on a Hydrophilic Acrylic Intraocular Lens: A Clinical Case Series and Material Comparative Study of Different IOLs. J Ophthalmol. 2019;2019(1):5746186.
18. Zhang S, Xu J. Impact of Patient Characteristics and Surgery-Related Risk Factors on Endophthalmitis after Cataract surgery: A Meta-Analysis. Ophthalmic Res. 2025;68(1):117-36.
19. Barry P, Cordoves L, Gardner S. ESCRS Guidelines for Prevention and Treatment of Endophthalmitis Following Cataract Surgery: Data, Dilemmas and Conclusions. Dublin, Ireland: European Society of Cataract and Refractive Surgeons; 2013.
20. Musumeci R, Troiano P, Martinelli M, Piovella M, Carbonara C, Rossi S, et al. Effectiveness of 0.66% Povidone-Iodine Eye Drops on Ocular Surface Flora before Cataract Surgery: A Nationwide Microbiological Study. J Clin Med. 2021;10(10):2198.
21. Wang XL, Huang XY, Wang Z, Sun W. The Anterior chamber injection of moxifloxacin injection to prevent endophthalmitis after cataract surgery: a meta-analysis. J Ophthalmol. 2020;2020(1):7242969.
22. Malmin A, Syre H, Ushakova A, Utheim TP, Forsaa VA. Twenty years of endophthalmitis: Incidence, aetiology and clinical outcome. Acta Ophthalmol. 2021;99(1):e62-9.
23. Yoon SJ, Kim SH, Bahk HJ, Ahn YS, Lee JJ, Kim HJ, et al. Fungal Endophthalmitis Outbreak after Cataract Surgery, South Korea, 2020. Emerg Infect Dis. 2022;28(11):2226-33.
24. Lalitha P, Sengupta S, Ravindran RD, Sharma S, Joseph J, Ambiya V, et al. A literature review and update on the incidence and microbiology spectrum of postcataract surgery endophthalmitis over the past two decades in India. Indian J Ophthalmol. 2017;65(8):673-7.
25. Gower EW, Keay LJ, Stare DE, Arora P, Cassard SD, Behrens A, et al. Characteristics of Endophthalmitis after Cataract Surgery in the United States Medicare Population. Ophthalmology. 2015;122(8):1625-32.
26. Naik AU, Prakash VJ, Susvar P, Therese KL, Parameswari CK. Postoperative endophthalmitis due to Pseudomonas luteola: first reported case of acute and virulent presentation from a tertiary eye care center in South India. Indian J Ophthalmol. 2018;66(8):1200-2.
27. Ben Hmida S, Boughariou I, Gassara F, Maazoun M, Eleuch E, Marrakchi C, et al. Pseudomonas Luteola Infection: first case report of urinary tract infection and review of literature. Electron J Gen Med. 2021;18(5):em313.
28. Awitan HLG, Jumalon NJU, Neria ACC, Pilarta CT, Suarez ECPL, Tinaja CJ, et al. Microbial Characterization of Healthcare-associated Infection-causing Bacteria Isolated from Alcohol Dispensers in a Tertiary Hospital in Manila. 2017. Available in: https://tuklas.up.edu.ph/Record/UP-1685523046126318462
29. Narnaware SH, Bawankule PK. Post-cataract surgery endophthalmitis caused by acinetobacter baumannii. Int J Contemp Med Surg Radiol. 2019;4(3):C38-C39.
30. Oh DH, Kim YC, Kim EJ, Jung IY, Jeong SJ, Kim SY, et al. Multidrug-resistant Acinetobacter baumannii infection in lung transplant recipients: risk factors and prognosis. Infect Dis (Lond). 2019;51(7):493-501.
31. Mikosz CA, Smith RM, Kim M, Tyson C, Lee EH, Adams E, et al; Fungal Endophthalmitis Outbreak Response Team. Fungal endophthalmitis associated with compounded products. Emerg Infect Dis. 2014;20(2):248-56.
32. Brooks RB, Mitchell PK, Miller JR, Vasquez AM, Havlicek J, Lee H, et al; Burkholderia cepacia Workgroup. Burkholderia cepacia Workgroup. Multistate outbreak of Burkholderia cepacia complex bloodstream infections after exposure to contaminated saline flush syringes: United States, 2016-2017. Clin Infect Dis. 2019; 69(3):445-9.
33. Southworth PM. Infections and exposures: reported incidents associated with unsuccessful decontamination of reusable surgical instruments. J Hosp Infect. 2014;88(3):127-31.
34. Park J, Popovic MM, Balas M, El-Defrawy SR, Alaei R, Kertes PJ. Clinical features of endophthalmitis clusters after cataract surgery and practical recommendations to mitigate risk: systematic review. J Cataract Refract Surg. 2022;48(1):100-12.
35. Kamali M, Manshouri S, Bagheri Y, Rostami M, Karkhaneh Mahmoudi M, Moradnejad P, Seif F. Prevalence and antibiotic resistance of Acinetobacter baumannii among patients in postcardiac surgery intensive care units of Rajaei Hospital, Tehran. Med J Islam Repub Iran. 2020;34:4.
Data Availability Statement: The datasets generated and/or analyzed during the current study are already available.
Edited by
Editor-in-Chief: Newton Kara-Júnior
Submitted for publication:
July 28, 2025.
Accepted for publication:
January 23, 2026.
Approved by the following research ethics committee: Universidade Federal de Rondônia (CAAE 20070719.5.0000.5300).
Funding: This study received no specific financial support.
Disclosure of potential conflicts of interest: The authors declare no potential conflicts of interest.